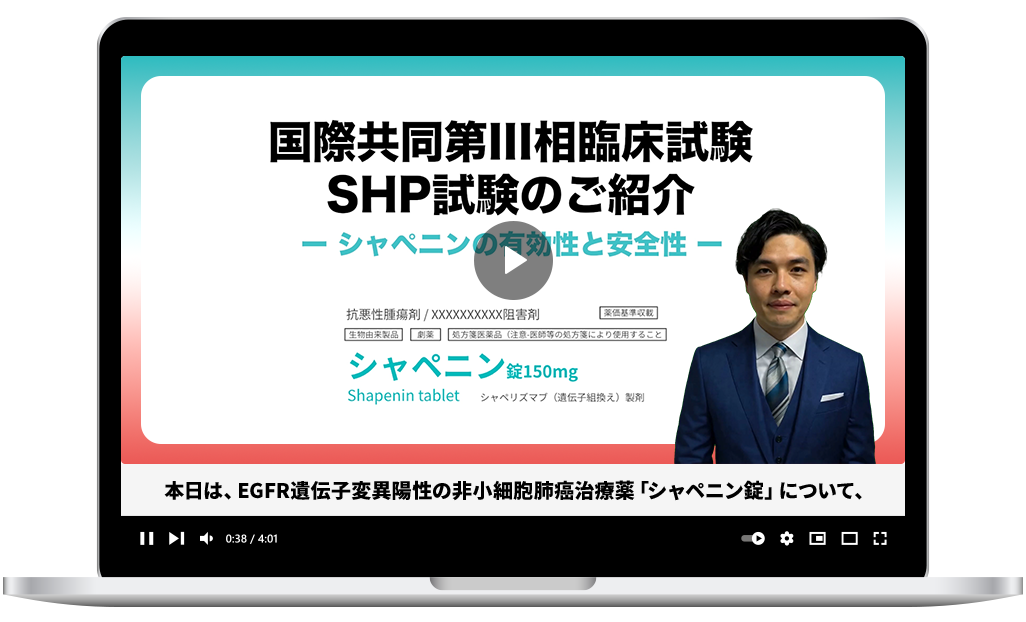
Create materials aligned with production guidelines in short lead times and at lower cost*1 *2. Expand your content lineup and shorten your promotional cycle.

It takes months from planning to delivery, making it hard to share congress updates and the latest evidence in a timely way.

External production costs are high, so budget constraints prevent broader content development and series expansion.

Drafting and compliance review require substantial effort, making it hard to stay focused on strategy.

Teams hesitate to adopt AI because they worry about misinformation and whether outputs will align with internal rules.
Simply upload your existing materials, such as PDFs or videos, and the AI will automatically produce and deliver digital content such as videos and HTML*1.
Submit your existing material data as-is



Choose the best content format based on the materials you already have.
Simply upload your product presentation slides and the workflow runs end-to-end, from script generation to narration and video production.


Automatically generate mobile-friendly HTML from leaflet PDFs and meeting-slide PDFs, greatly reducing production work for multichannel deployment.

Create content from event materials in formats such as summary reports, digest videos, and email templates.


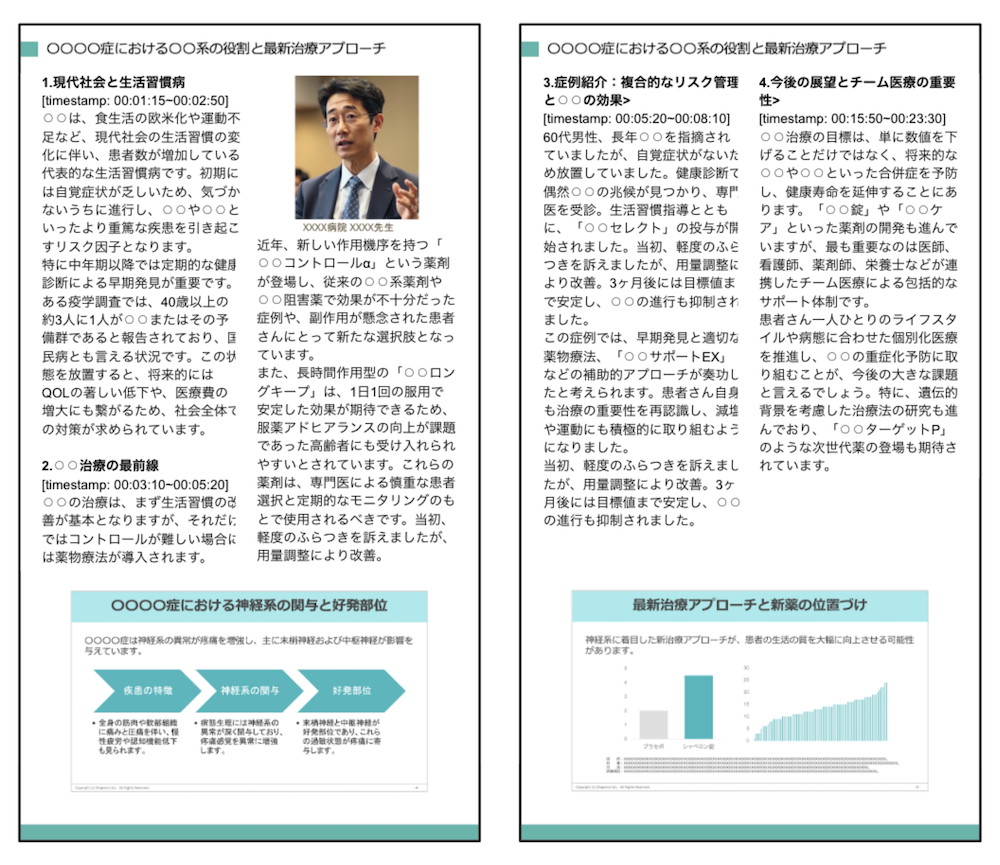
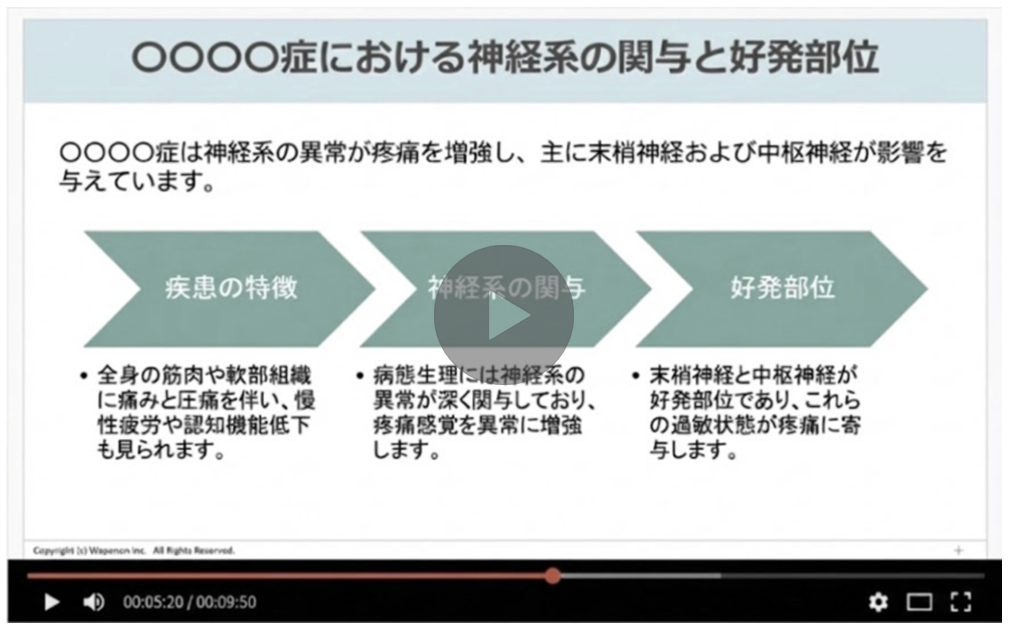
Using a single existing asset, AI creates slides, videos, and other content optimized for physician segments such as conservative or proactive prescribers, making personalized rollout more efficient.

 Conservative prescribers
Conservative prescribers
 Proactive prescribers
Proactive prescribers
Your existing slides can reach this level
of quality in as little as 5 business days*3.
Compared with conventional outsourcing, content can be produced at lower cost. That opens up projects such as FAQ videos and disease-awareness content that may previously have been out of reach.
Share time-sensitive information such as regulatory changes and new product updates quickly, and get it to physicians ahead of competitors.
Draft content based on pharmaceutical advertising guidelines. Adjustments for company-specific rules*5 are also possible, reducing workload for both production and review.
Turn congress slides into videos and HTML right away, so content where freshness matters can reach physicians ahead of competitors.

Create summaries and digest videos immediately after events. MRs can quickly catch up on the lecture content and follow up with physicians promptly and effectively.

Turn existing slides and materials, such as disease-awareness content, into videos. Narration and animation help make them easier to understand while expanding site content with less effort.

Contact Us
Please contact us first and tell us about your challenges and the kinds of content you want to create.
Align on the production approach
We review your source data, target physician audience, production goals, and desired options, then align on the content production approach.
Draft production and revision
We prepare the first draft based on the agreed production approach, reflect revision requests, and address feedback after review by your internal approval team.
Delivery
We deliver the final version.
Not limited to the services shown on this page,
you can consult us about any content-production challenge.